To start a GLOBALG.A.P. Compound Feed Manufacturing v3.1 audit, we need to create our certification scope. To do so, we first must create the producer’s product scope site. We start by selecting the Type “Sites/PHU” and afterwards one of the applicable "Sites/PHU and Category "GLOBALG.A.P. Compound Feed Manufacturing - Scope Registration (Site)":
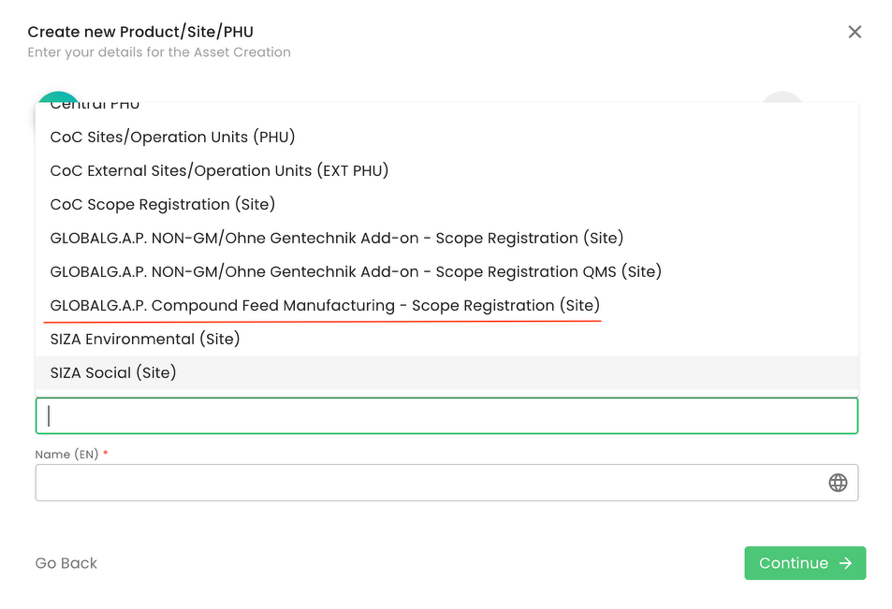
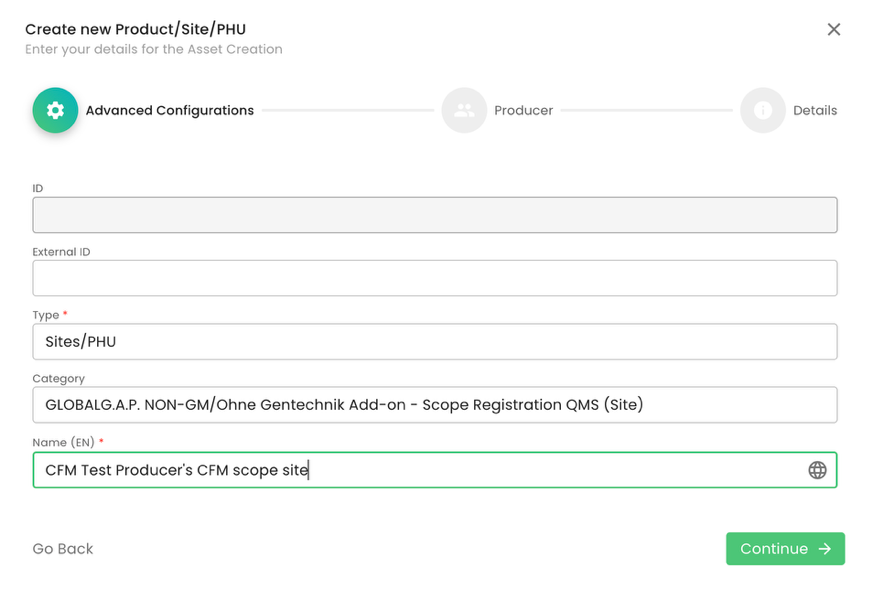
As per standard practice, proper site naming is essential:
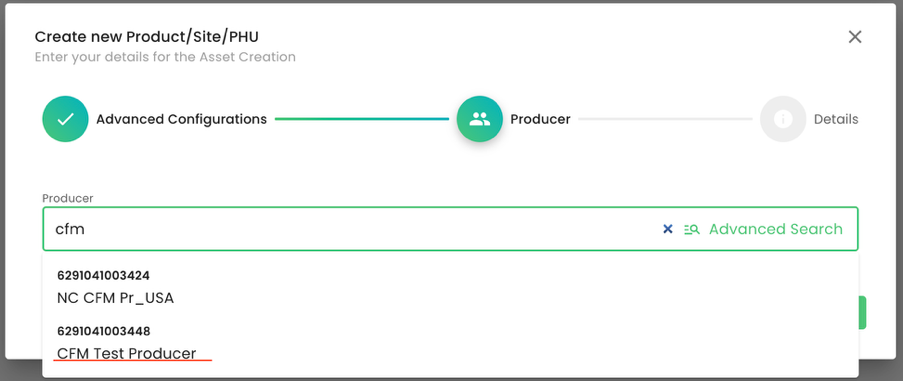
As always, we select the producer. In this example, it is the producer “CFM Test Producer”:
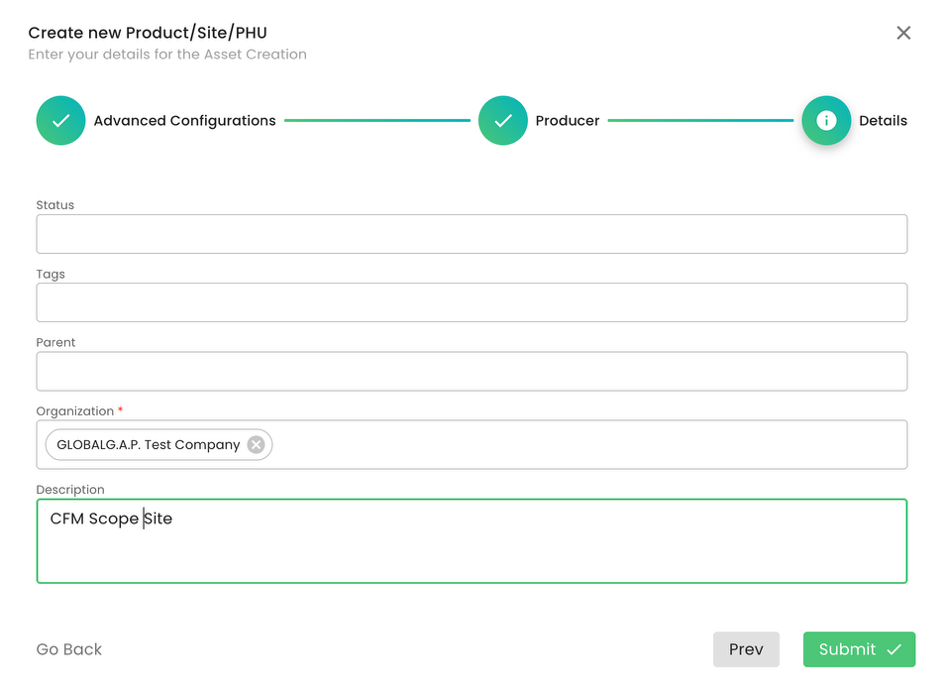
Please note that adding a contact person is not required.
We do not add a Parent; we select our CB under organization and submit. As always, you can add tags or a description to the respective fields. Please note that for the scope site, an address is not required:
The next step is to add the certification scope (product) to the site above.
The available products for this FAP are:
“Compound Feed for Aquaculture” (ID: 11189) &
“Compound Feed for Livestock” (ID: 11190)
As always, we select Type “Product” and properly name it. In our example, the certification scope is “Compound Feed for Aquaculture”:
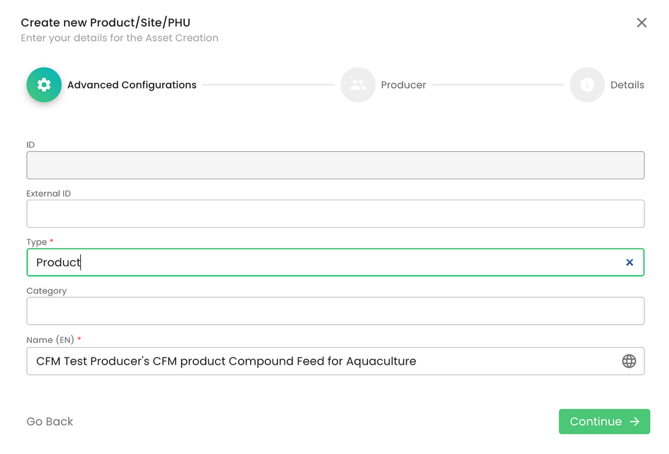
We select the producer:
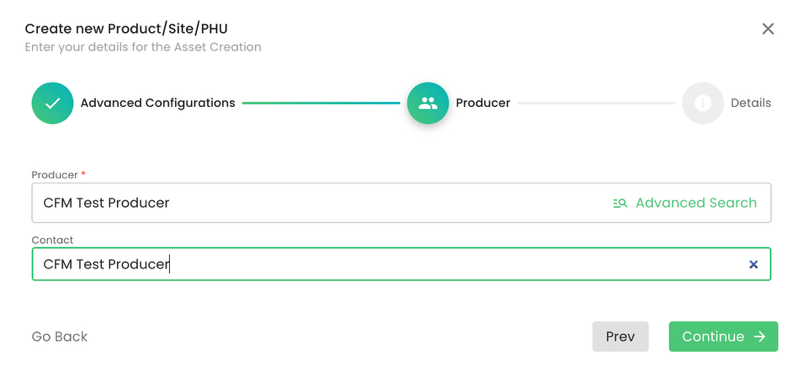
In our example, we will select the product “Compound Feed for Aquaculture” (ID: 11189), select under Parent the site we have created before, add our CB under Organization and press on submit:
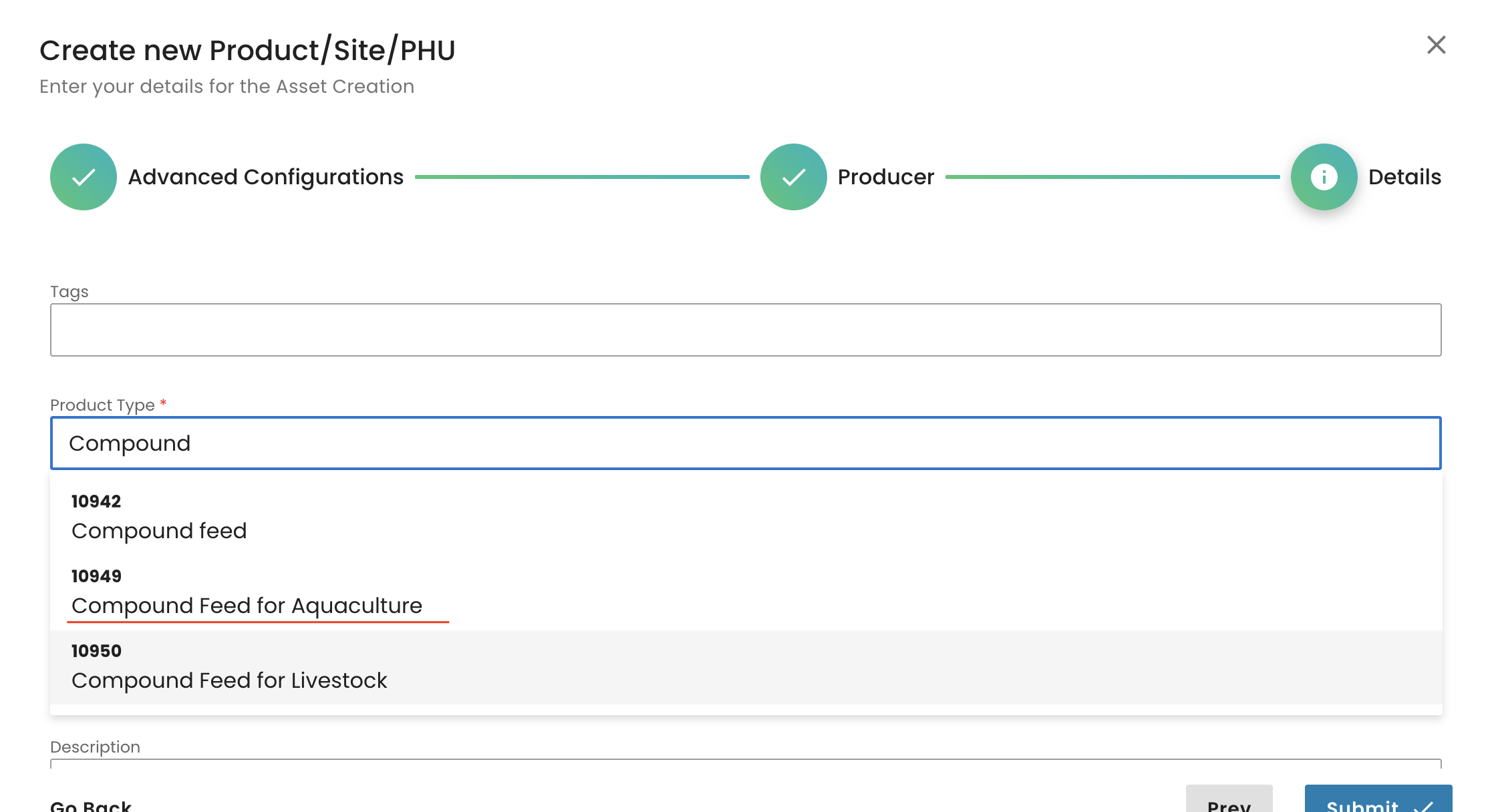
We select the parent and submit:
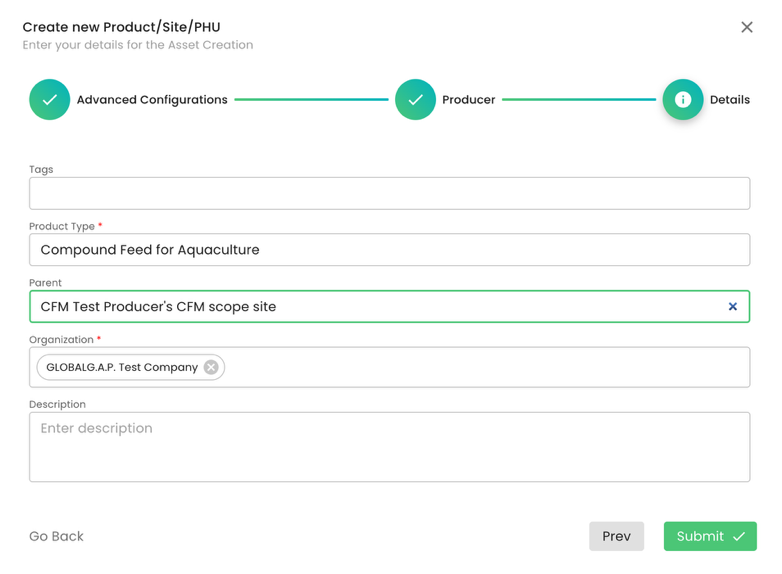
Note: This standard does not require a PHU.
To summarise, in our example, we have created,
Please note that the creation of the scope site and product scope needs to be done only for the first audit and can be used for all future audits.
After having finalised the creation of the scope, we can start the audit in the same way as always by selecting the Farm Assurance Product (FAP). First, we select the relevant FAP and the applicable reporting option:
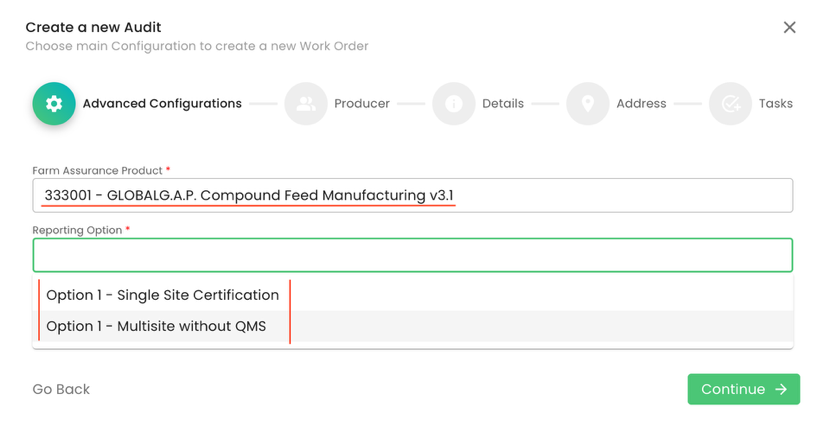
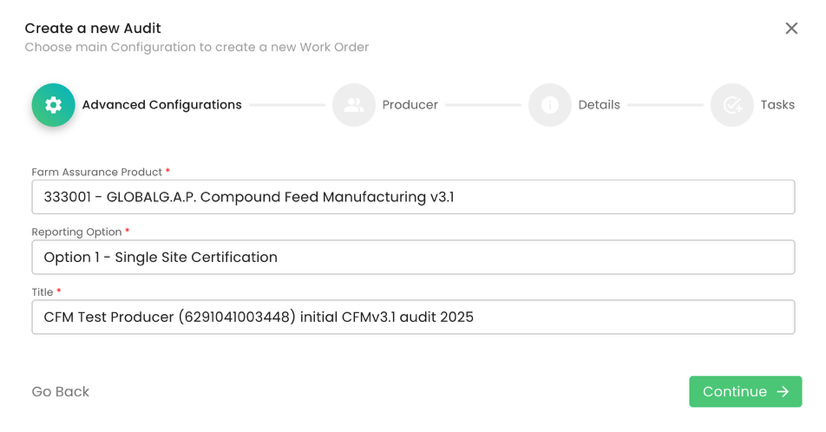
In our scenario, we select Option 1 - Single Site Certification. Proper audit naming is essential:
Please note: For Option 1 Multisite without QMS, kindly select the appropriate reporting option and refer to the information provided following the "Smartforms screenshot" below.
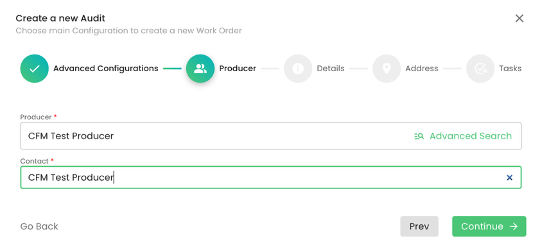
We select the producer and contact person:
We select our CB, and under Audit type, we select the applicable option. In our example, we selected “Initial certification audit”.
Note: for all GLOBALG.A.P. Compound Feed Manufacturing v3.1 audits, under Audit Product Category, we shall select “GLOBALG.A.P. Compound Feed Manufacturing scope”:
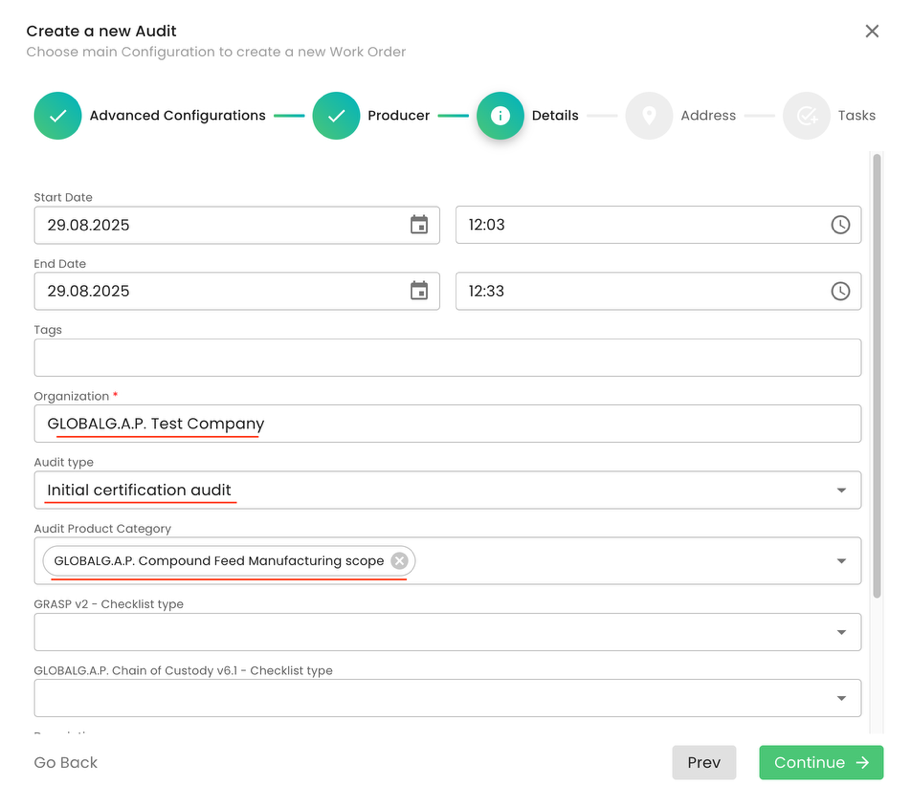
We click through the pop-up window by clicking “Continue” and “Submit” the audit.
The next step is to add the Scope information to our audit:
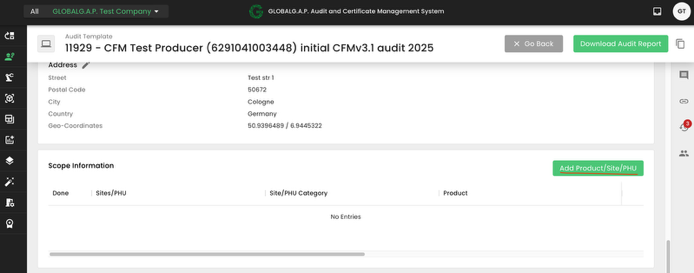
&

With this, the proper smart forms will be triggered:
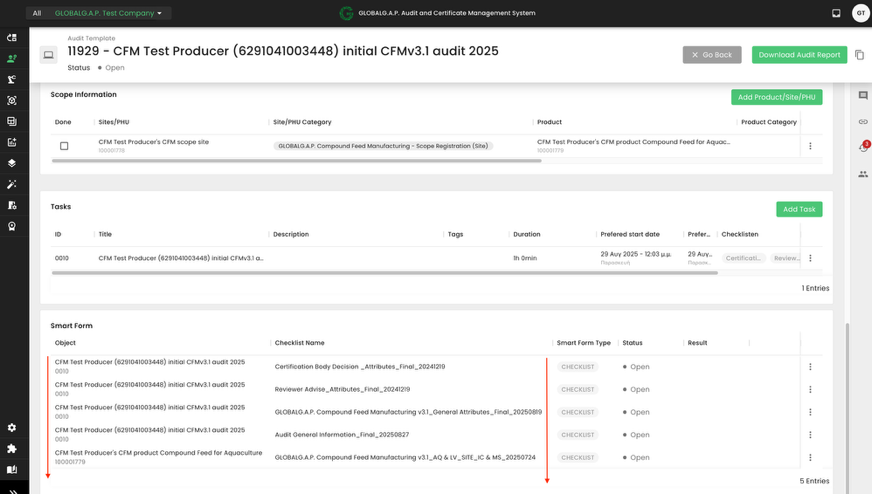
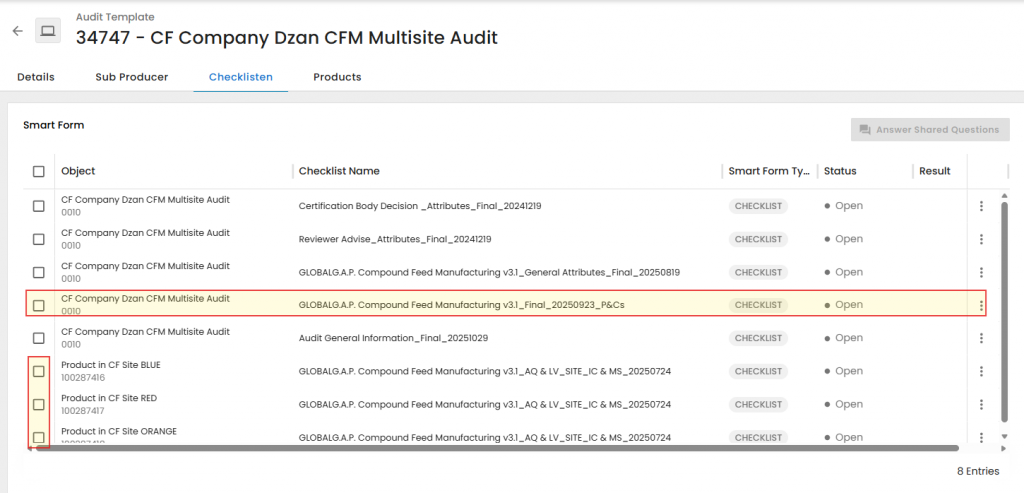
Option 1 Multisite Without QMS:
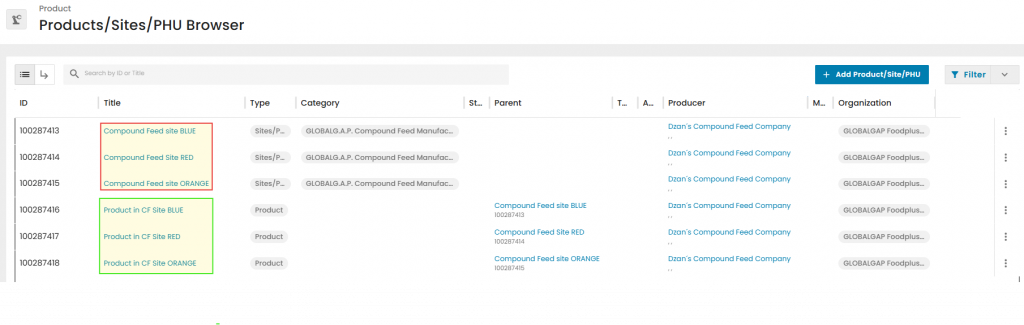
If there are several production sites, CB must register each site for every product. This means the same CFM site (GLOBALG.A.P. Compound Feed Manufacturing - Scope Registration (Site)) may appear multiple times with different names or products, similar to IFA registration for multisite producers.
Next, add created Compound Feed Scope Products from registered CF Scope Sites to the Audit under "Products" list.
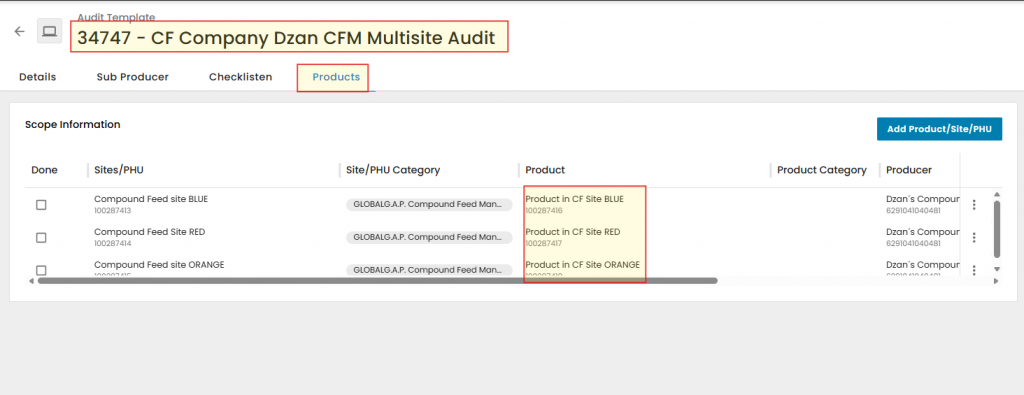
Please note that during the creation of this wiki, the P&C smart form was not available. This is the reason it is not displayed in the above screenshot for Option 1- Single Site certification.
The P&C smart form is displayed below for Option 1 - Multisite Without QMS audit, which was created later along with the relevant Product Attribute smart form for each site.
After filling out the smart forms and finalizing the audit, the certificate can be issued as explained in this wiki.